imuno® is 100+ times more potent than pure GcMAF
Superior Immune & Mitochondrial Support
- Rationale for the design of a novel tool for immunotherapy. Ruggiero M, Pacini S. Switzerland 27 Aug 2018, Integrative Cancer Science and Therapeutics, pages 1-5.
RESEARCH ARTICLE
- A Novel potential Adjuvant for Cancer Vaccines. Ruggiero M, Pacini S. 29 Sept 2018, Madridge Journal of Vaccines, Vol. 2, pages 58-62.
RESEARCH ARTICLE
- Clinical Experience of Integrative Autism treatment with a Novel type of Immunotherapy. Antonucci N, Pacini S, Ruggiero M. Italy 22 Feb 2019, Madridge Journal of Vaccines, Vol. 3, pages 71-76.
RESEARCH ARTICLE
- Tailoring the Ruggiero-Klinghardt Protocol to Immunotherapy of Autism. Antonucci N, Klinghardt D, Pacini S, Ruggiero M. 24 Apr 2019, American Journal of Immunology, Vol. 14, pages 34-41.
RESEARCH ARTICLE
- Use of an Extremely Biodiverse Probiotic and a Supplement Based on Microbial Chondroitin Sulfate is Associated with a Significant Decrease of Serum Free Kappa Light Chains as well as a Trend Toward Normalization of Kappa/Lambda Ratio and of Plasma Cell Bone Marrow Infiltration in a Case of Multiple Myeloma. Antonucci N, Pacini S, Ruggiero M. 18 Jun 2019, American Journal of Immunology, Vol. 15, pages 5-9.
RESEARCH ARTICLE
- Color Doppler Evaluation of Isovolumetric Relaxation Time and of Signals Arising from Axons of the Median Nerve as a Means to Evaluate Mitochondrial Functionality in the Context of Immunotherapy of Cancer and Chronic Conditions Associated with Mitochondrial Dysfunction. Stefania Pacini and Marco Ruggiero. 12 Oct 2019, American Journal of Immunology.
RESEARCH ARTICLE
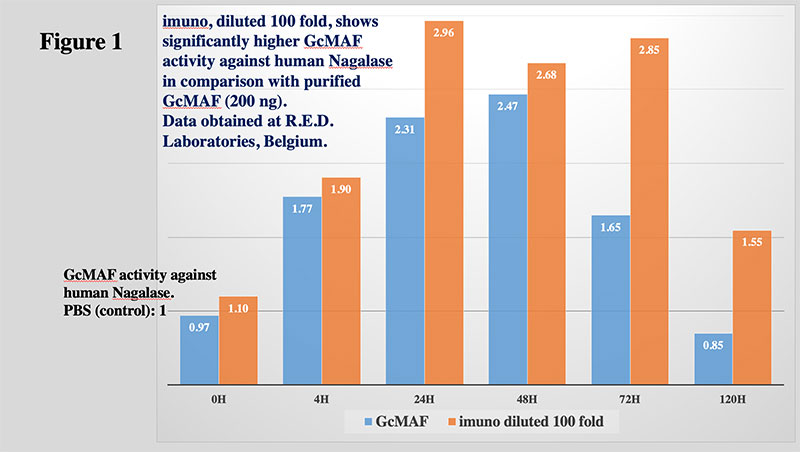
- imuno® is over 100 times more effective than pure GcMAF. Marco Ruggiero. 19 Feb 2020, Public Letter.
RESEARCH ARTICLE
- Consumption of an Extremely Biodiverse Probiotic and a Supplement based on Microbial Chondroitin Sulfate is Associated with Very Low Serum Alpha-N-acetylgalactosaminidase (Nagalase) Activity and Decrease of C-reactive Protein Values. Michael Carter, Stefania Pacini and Marco Ruggiero. 20 March 2020, American Journal of Immunology.
RESEARCH ARTICLE
imuno® is a standardized, homogenous, ultrapure, supramolecular complex constituted by ultrapure phosphatidylcholine of non-animal origin, homogeneous low-molecular-weight chondroitin sulfate of non-animal origin, and vitamin D3. Complexing the three active molecules in one single structure enables activation of the receptors for each molecule simultaneously in a number of cells, thus amplifying by multiplication the efficacy of each molecule taken singularly. In other words, imuno® has all the pharmacological properties of vitamin D3, phosphatidylcholine, and chondroitin sulfate combined and amplified.
The receptors for the components of imuno® are present on the surface and in the nucleus of all the cells of the immune system. The components of imuno® activate their receptors, simultaneously, on all the cells of the immune system, leading to regulation of gene expression in the nucleus and the mitochondria, increased production of energy in the mitochondria, and a shift toward non-inflammatory activation of the immune system. Listed below are the activation of cells of the immune system based on peer-reviewed scientific literature.
- Activates Macrophages.
- Has Anti-inflammatory Effects.
- Promotes Macrophage M1 Polarization.
- Modulates the Immune System through Dendritic cells.
- Increases the Activity of Natural Killer cells.
- Activates Neutrophils.
- Modulates T-cell-mediated Immunity.
- Increases Mitochondrial Energy.
- Provides all GcMAF Functionality.
This website is accessible from many geographical locations, and the information that it provides regarding imuno® is not applicable to, nor intended to satisfy the requirements of all countries. imuno Corporation supplies imuno® to distributers throughout the world and does not make any claims regarding the use of these products and each distributor is responsible for ensuring that the claims made for and use of imuno® comply with the regulatory requirement of the locations in which it markets these products.
- GcMAF Made Better. (imuno® is 100+ times more potent than pure GcMAF)
- Superior Immune Support (BE WELL).
- Superior Endocannabinoid Receptor Support (FEEL WELL).
- Superior Mitochondrial Support (HAVE ENERGY).
- Superior Lymphatic Drainage Support (A BETTER FLOW).
imuno® is designed by Molecular Biologist, Dr Marco Ruggiero, MD. PhD. Author of peer-reviewed scientific papers on immunotherapy and quantum biology.
imuno® is produced under licence in a pharmaceutical facility which is audited by Medsafe, a division of the Ministry of Health of New Zealand, to both GMP and Pharmacy Standards NZ.
imuno® is the elegant solution. It is the revolutionary supra-molecular structure that reproduces the self-assembly of cell components. This delivers the combined molecules, at the same time, at the required dose, to their biologic targets, and produces powerful support. Also, the manufacturing process for imuno® is meticulous and time-consuming, and it is not technically possible to get the same result by simply mixing the active ingredients together since the procedure follows a proprietary algorithm based on biologic negentropy.
imuno Distribution.
Online SHOP
Natural Solutions NZ Ltd.
103 Hugo Johnston Drive,
Penrose, Auckland 1061,
New Zealand.
sales@naturalsolutions.nz
+64-9-3681909
https://naturalsolutions.nz
Click me to buy imuno online
BioPlus Health Center
Unit A01-01, No.1, Harvard St,
Diamond Island, Tonle Basac, Chamkarmon,
Phnom Penh, Cambodia 12300.
bioplus@protonmail.com
+85-517771088
Totech Shanghai.
Room 1410 Block B, No.18, Taolin Road,
Shanghai 200135, P.R.China.
totechshanghai@163.com
+86-021-33820932
Totech Japan Ltd.
1-2-5F United Asian, Tomiyama-cho,
Kanda, Chiyoda-ku, 101-0043 Japan.
info@totech.com
+81-3-6811-6201
CTC Life Sciences Inc.
#507 Mario Tower, 28, Digital-ro, 30-gil,
Guro-gu, Seoul, Rep.of Korea, 08389
ysyyoung64@naver.com
+82-2-867-8823
Root of Life.
53 Kim Keat Road, #03-03,
Singapore 328823.
contact@rootoflife.co
https://rootoflife.co
Click me to buy imuno online
Root of Life.
53 Kim Keat Road, #03-03,
Singapore 328823.
contact@rootoflife.co
https://rootoflife.co
Click me to buy imuno online
YourHealthBasket.
Drakes Lane Industrial Estate,
Unit 1 Drakes Lane,
Boreham Essex CM3 3BE,
United Kingdom.
mail@yourhealthbasket.co.uk
+44-1245-905505
https://www.yourhealthbasket.co.uk
Click me to buy imuno online
Oxygen Health Systems LLC.
2154 Oak Ct
Naperville, Illinois 60565,
United States.
oxygenhealthsystems@gmail.com
+1-630-451-8821
https://www.oxygenhealthsystems.com
Click me to buy imuno online
Healthy Energetics LLC.
La Cita Area,
Titusville, Florida 32780,
United States.
orderdesk@healthyenergetics.com
+1-321-747-5517
https://www.healthyenergetics.com
Click me to buy imuno online

